- Blog
- Download d3dx11
- Black axe confraternity
- The queen singapore drama
- Racer sideways slot car riley
- Ptv home
- Autodesk student license
- Stronghold legends steam edition
- Valkyrie drive ed
- Kotor 2 plague carrier
- Core 2 quad q8200 review
- Download ps1 bios file
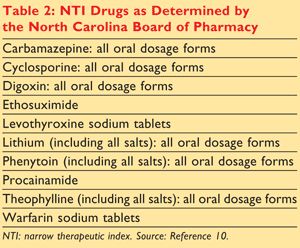
- Therapeutic index range
- Sims 2 food cc
- How to check pc temps windows 10
- Hollow knight overcharmed
Autoradiography, MALDI-MS, and SIMS-MS imaging in pharmaceutical discovery and development. Pharmacokinetics and pharmacokinetic/pharmacodynamic relationships for angiotensin-converting enzyme inhibitors. The minimum anticipated biological effect level (MABEL) for selection of first human dose in clinical trials with monoclonal antibodies. α4β2 neuronal nicotinic receptor positive allosteric modulation: an approach for improving the therapeutic index of α4β2 nAChR agonists in pain. Recognition of adverse and nonadverse effects in toxicity studies. The effect of plasma protein binding on in vivo efficacy: misconceptions in drug discovery. (eds) Lippincott's Illustrated Reviews: Pharmacology 4th edn (Lippincott Williams & Wilkins, 2008). Tissue-specific, non-invasive toxicity biomarkers: translation from preclinical safety assessment to clinical safety monitoring. Proteomics in the search for mechanisms and biomarkers of drug-induced hepatotoxicity. Van Summeren, A., Renes, J., van Delft, J.

Genomic and proteomic approaches for predicting toxicity and adverse drug reactions. Use of transcriptomics in understanding mechanisms of drug-induced toxicity. In silico models for drug-induced liver injury - current status. Drug safety sciences and the bottleneck in drug development. Permitting product liability litigation for FDA-approved drugs and devices promotes patient safety. The role of litigation in defining drug risks. Beyond debacle and debate: developing solutions in drug safety.
#THERAPEUTIC INDEX RANGE HOW TO#
How to improve R&D productivity: the pharmaceutical industry's grand challenge.
